Bioapplication
Biointerface, LNP-Relevant, and Therapeutic Nanomaterials
A project area connecting lipid-coated nanomaterials, protein-mediated targeting, analytical characterization, and biologically relevant assay systems with direct relevance to drug delivery, biologics protection, and therapeutic platform development.
Overview
This project sits in a part of research that I believe is highly relevant to biotechnology and pharmaceutical development: how to design nanomaterial systems that remain stable and biocompatible under mild conditions, but become functional when they are brought into the right biological context. In this specific case, the system was designed for selective viral inactivation, but the broader logic is closer to the way I think about therapeutic and delivery platforms more generally: controlled activation, targeted interaction, and analytically tractable performance in biologically relevant environments.
The case study shown here involved a silver nanoparticle core, a passivating lipid coating, the photocatalyst [Ru(bpy)3]2+, and annexin V as a targeting ligand for phosphatidylserine on enveloped viruses. Under 470 nm illumination, the system generated reactive oxygen species and increased the permeability of the lipid shell, enabling localized photoreactivity and Ag+ release near the bound virus. Using murine leukemia virus (MLV) as a model, annexin V-functionalized plasmonic nanoreactor virucides achieved over 85% viral inactivation after 30 minutes of illumination at a 1:1 virus-to-particle ratio with no measurable cytotoxicity in host cells.


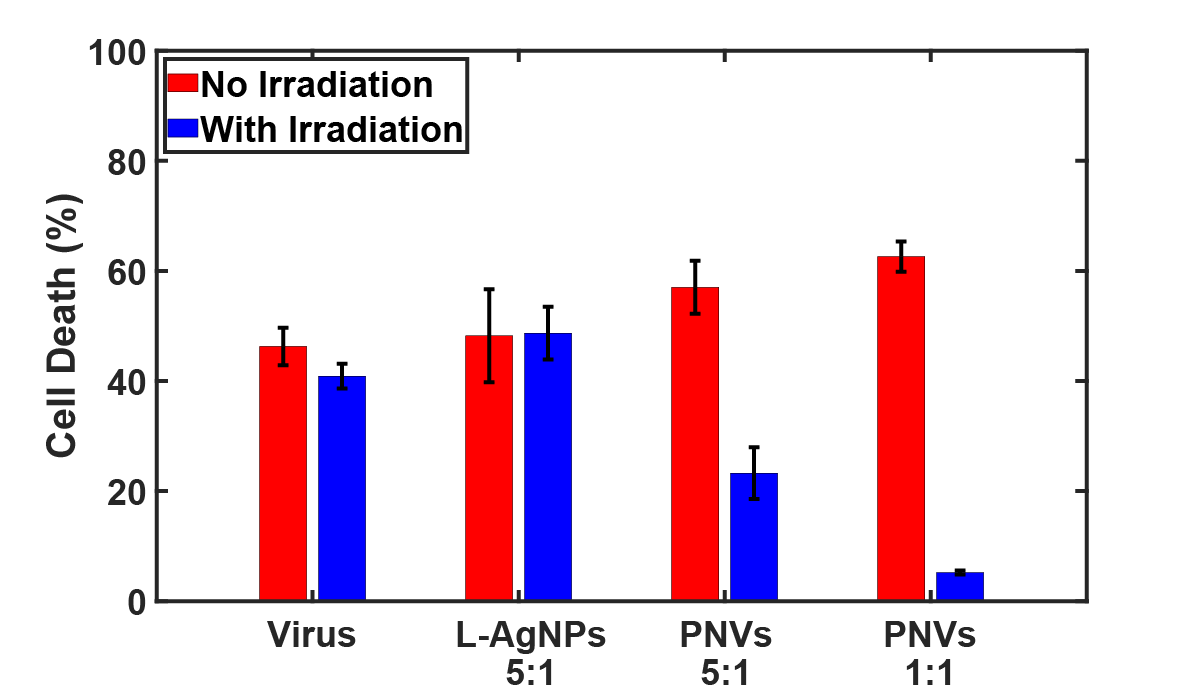
What I Worked On
- Helped develop lipid-coated silver nanoparticle hybrids that localize [Ru(bpy)3]2+ in the evanescent plasmonic field.
- Worked on nanoparticle functionalization logic, including azide-bearing lipids and annexin V targeting chemistry.
- Contributed to the characterization of structure and composition through TEM, STEM-EDX, UV-vis, hydrodynamic size, and zeta potential measurements.
- Studied how resonant illumination drives ROS generation, lipid-shell destabilization, and increased Ag+ accessibility.
- Integrated virology-facing assays to evaluate viral inactivation, host-cell viability, and the role of targeting in improving selectivity.
Broader Biochemistry and Pharma-Relevant Experience
Beyond this specific nanoreactor system, my broader experience is relevant to biotech and pharma-facing work that involves lipid-based materials, protein interactions, analytical characterization, and biologically relevant assays.
- Interested in LNP-relevant systems, targeted delivery logic, and formulation behavior under biologically relevant conditions.
- Comfortable thinking across chemistry, biochemistry, analytical science, and assay design rather than treating them as separate domains.
- Interested in problems where material performance must be evaluated in ways that are useful for therapeutic development and biopharmaceutical research.
Analytical and Experimental Strengths
- Experience with analytical and bioanalytical characterization, including UV-vis, HPLC, FT-IR, mass spectrometry, ICP-MS, and MP-AES.
- Hands-on work with nanoparticle characterization methods such as TEM, SEM, AFM, and optical imaging.
- Experience designing and troubleshooting assays in biologically relevant environments rather than only idealized materials settings.
- Familiarity with protein-related workflows, biointerface engineering, and quantitative evaluation of nanoparticle-biomolecule interactions.
- Comfort working with lipid-based systems where stability, targeting, permeability, and functional response all matter at the same time.
- Strong interest in how these tools can support LNP, therapeutic delivery, biologics protection, and AI-enabled pharmaceutical R&D.
Mechanistic Logic
What makes this system especially interesting is that it works through multiple coupled mechanisms rather than a single antiviral effect. The lipid shell initially passivates the silver nanoparticle and suppresses premature Ag+ release, which improves cytocompatibility under dark conditions. Upon resonant illumination, plasmon-enhanced excitation of the Ru photocatalyst generates ROS in the immediate vicinity of the virus. Those reactive species can damage the virus directly while also promoting lipid peroxidation in the nanoparticle coating, which increases shell permeability and enables additional Ag+ release near the target.
Annexin V adds an important targeting layer by binding phosphatidylserine exposed on enveloped viruses, allowing reactivity to be concentrated where it is most useful. In other words, the project is not just about making a reactive nanoparticle, but about combining targeting, controllable activation, and local chemistry in a way that improves efficacy while minimizing collateral damage.
Why It Matters
- This project connects lipid-based interfaces, protein-mediated targeting, assay logic, and analytical characterization in one experimentally grounded system.
- It reflects the kind of work I want to do in biotech and pharmaceutical settings, where translation depends on understanding both platform design and biological context.
- It also shows that I can contribute not only to materials questions, but to formulation, targeting, and therapeutic-relevance problems more broadly.